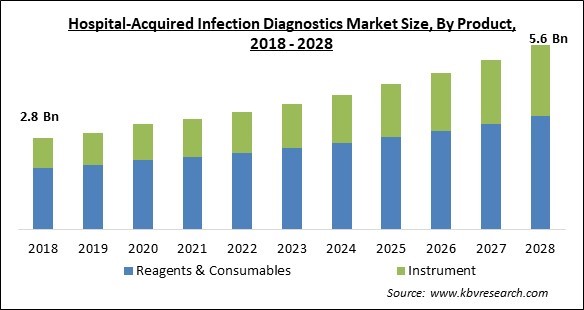
The Global Hospital-Acquired Infection Diagnostics Market size is expected to reach $5.6 billion by 2028, rising at a market growth of 7.8% CAGR during the forecast period.
Infections acquired in hospitals are associated with viral, bacterial, or fungal pathogens. Numerous infections can be obtained in clinical settings such as nursing homes, rehabilitation center, outpatient clinics, or other therapeutic settings. It can be acquired from an infected patient, the surrounding environment, or the facility's staff. All patients are at risk for infections acquired in the hospital.
People that had surgery, the children and the elderly, are more likely to get hospital-acquired infections. The most common infections are surgical-site infections, respiratory infections, urinary tract infections, gastrointestinal infections, cardiovascular system infections, bloodstream infections, and bone and joint infections. Hospital-acquired infections (HAIs) are a leading cause of death among hospitalized patients in both developed and emerging countries.
All across the world, the amount of surgical procedures conducted each year has been steadily increasing, and by the end of the decade, it is predicted to hit a new high. As the need for surgical instruments grows along with the number of surgical procedures performed, the risk of healthcare-associated infections is expected to rise. A growing understanding of the value of sterilized and disinfected medical tools and devices would help drive demand for nosocomial infection control equipment. Other factors driving sales of hospital obtained infection control systems include the increased frequency of chronic diseases and advances in surgical equipment.
Many clinicians can identify a HAI (Hospital acquired infection) based just on appearance and symptoms. Inflammation and/or a rash at the infection site might also be signs. HAIs do not include infections that develop complex prior to the stay. However, any additional symptoms should be reported to the doctor during the stay. The treatment for various illnesses is determined by the type of infection. Antibiotics and bed rest are likely to be prescribed by the doctor. It will also remove any foreign devices like catheters as soon as it's medically necessary.
COVID-19 was labeled a pandemic and overall well-being crisis of international concern by the World Health Organization (WHO) on January 30, 2020. Around 210 countries have been affected by COVID-19. Governments around the world have imposed mandatory lockdowns and social distancing to prevent the healthcare system from collapsing. Projects and elective procedures have also been subjected to limits and restrictions by states. The roadblocks continue to impede the expansion of various industries, and the increased response to the rules has an impact on the industries. Lockdowns around the world have damaged the distribution, production, and retail networks.
Except for bacterial spores from inanimate things, disinfection is a technique that destroys the majority or all pathogenic microorganisms. Wet pasteurization or liquid chemicals are used to accomplish it. Several factors have influenced disinfection efficiency, each of which might limit the process' efficacy. The use of disinfectants in healthcare facilities to protect people doing the medical process that exposes people to the virus is on the rise. During the COVID-19 pandemic, the need for surface disinfectants was huge and would grow significantly in the coming years.
The amount of surgical treatment has increased dramatically throughout the years. Every year, many main surgical procedures, are conducted all over the world. The increased incidence of obesity and other lifestyle disorders, the growing senior population, and the rising prevalence of spinal injuries and sports-related injuries are all contributing to the rising cases of surgical procedures. As the amount of surgical treatment performed grows, so does the demand for various surgical equipment and medical gadgets.
To save money and avoid waste, healthcare practitioners reprocess reusable medical devices. Surgical forceps, stethoscopes, and endoscopes are instances of reusable medical devices. Although, it has questions regarding the reprocessed devices' safety and functionality. Improper instrument cleaning and sterilization can lead to the retention of blood, tissue, and other biological waste in reusable devices, raising the risk of surgical-site infections (SSIs) in patients. Microbes may be able to resist disinfection or sterilization because of the debris, which could contribute to HAIs.
Based on Product, the market is segmented into Reagents & Consumables and Instrument. The reagents and consumables segment acquired the highest revenue share in the hospital-acquired infection diagnostics market in 2021. This is due to low awareness and a higher incidence of hospital-caused infections in low and medium-income nations. Chemicals and biological agents are employed in laboratory testing to detect or quantify an analyte, or the substance being evaluated or assessed. Cost, stability, availability, cold- or cool-chain requirements, and the risks associated with each variety vary greatly. Consumables are objects that are only utilized once during a test and then discarded and consist of items like coverslips and microscope slides that are specific to the test. Other consumables, which include alcohol, bleach, and gloves, are used throughout all testing services and are categorized as generic laboratory consumables.
Based on Infection Type, the market is segmented into Urinary Tract Infections (UTI), Hospital Acquired Pneumonia, Bloodstream Infections, Surgical Site Infections, Gastrointestinal Infections, and Others. The hospital-acquired pneumonia segment garnered a significant revenue share in the hospital-acquired infection diagnostics market in 2021. Hospital-acquired pneumonia (HAP), also known as nosocomial pneumonia, is a lower respiratory infection that was not incubated when the patient was admitted to the hospital and manifests clinically 2 or more days later. Early-onset pneumonia should be considered community-acquired pneumonia.
Based on Application, the market is segmented into Drug-Resistance Testing and Disease Testing. The drug-resistance testing segment garnered the highest revenue share in the hospital-acquired infection diagnostics market in 2021. This is because drug-resistance testing can be used to see if a drug-resistant virus was spread. When resistance test results are available, the initial regimen can be tweaked if necessary. It is due to the quick turnaround time, reduced cost, and greater sensitivity for detecting mixes of wild-type and resistant virus, genotypic testing is recommended over phenotypic testing.
Based on Test Type, the market is segmented into Molecular Diagnostics, Immunoassay, and Urinalysis. The Immunoassay segment procured a significant revenue share in the hospital-acquired infection diagnostics market in 2021. This test type is in demand due to an increase in infectious diseases, cancer, and pandemic outbreaks. Immunoassay is a highly selective bio-analytical approach that uses an antibody or antigen as a bio-recognition agent to quantify the existence or concentration of analytes ranging from tiny molecules to macromolecules in a solution.
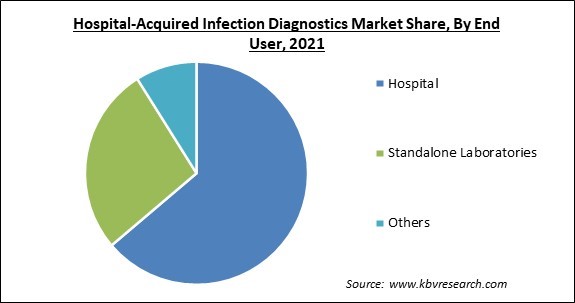
Based on End User, the market is segmented into Hospital, Standalone Laboratories, and Others. The hospital segment garnered the highest revenue share in the hospital-acquired infection diagnostics market in 2021. It is owing to the outbreak of a pandemic and a massive influx of patients. With the growth of new types of disease and the frequency of contraction of hospital-acquired infection, the role of hospitals comes into greater significance and will always grow in a progressive manner.
| Report Attribute | Details |
|---|---|
| Market size value in 2021 | USD 3.3 Billion |
| Market size forecast in 2028 | USD 5.6 Billion |
| Base Year | 2021 |
| Historical Period | 2018 to 2020 |
| Forecast Period | 2022 to 2028 |
| Revenue Growth Rate | CAGR of 7.8% from 2022 to 2028 |
| Number of Pages | 342 |
| Number of Tables | 543 |
| Report coverage | Market Trends, Revenue Estimation and Forecast, Segmentation Analysis, Regional and Country Breakdown, Competitive Landscape, Companies Strategic Developments, Company Profiling |
| Segments covered | Product, Infection Type, Application, Test Type, End User, Region |
| Country scope | US, Canada, Mexico, Germany, UK, France, Russia, Spain, Italy, China, Japan, India, South Korea, Singapore, Malaysia, Brazil, Argentina, UAE, Saudi Arabia, South Africa, Nigeria |
| Growth Drivers |
|
| Restraints |
|
Based on Regions, the market is segmented into North America, Europe, Asia Pacific, and Latin America, Middle East & Africa. North America emerged as the leading region in the hospital-acquired infection diagnostics market with the largest revenue share in 2021. It is due to a rise in the older population and the use of modern diagnostic equipment. As an outcome of the growing emphasis on healthy lifestyles and disease prevention among consumers, the market in this region is characterized by increasing demand for and adoption of sterilization and disinfection goods and services. The rise in the elderly population in the future years, as well as the requirement for hospitals, contracted infection control to reduce the prevalence of HAIs, and the execution of favorable government initiatives and stringent sterilization & disinfection regulations, are all driving the hospital-acquired infection diagnostics market in the region.
Free Valuable Insights: Global Hospital-Acquired Infection Diagnostics Market size to reach USD 5.6 Billion by 2028
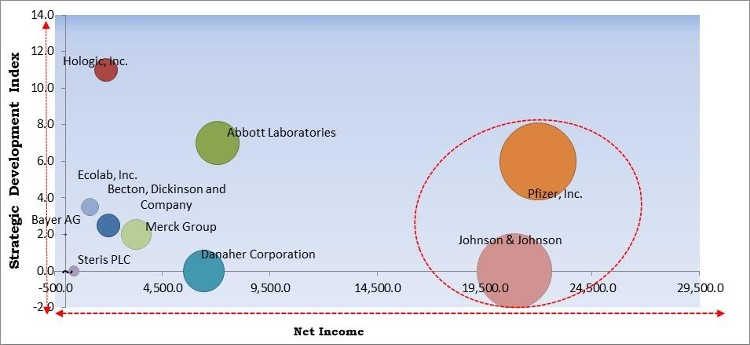
The major strategies followed by the market participants are Product Launches. Based on the Analysis presented in the Cardinal matrix; Johnson & Johnson and Pfizer, Inc. are the forerunners in the Hospital-Acquired Infection Diagnostics Market. Companies such as Abbott, Becton, Dickinson and Company, Ecolab, Inc. are some of the key innovators in the Market.
The market research report covers the analysis of key stake holders of the market. Key companies profiled in the report include Bayer AG (Bayer Schering Pharma AG), Danaher Corporation, Becton, Dickinson and Company, Hologic, Inc., Ecolab, Inc., Pfizer, Inc., Steris PLC, Johnson & Johnson, Abbott Laboratories, and Merck Group.
By Product
By Infection Type
By Application
By Test Type
By End User
By Geography
The global hospital-acquired infection diagnostics market size is expected to reach $5.6 billion by 2028.
The growing number of the surgical procedure are increasing are driving the market in coming years, however, problems associated with the safety of reprocessed medical instruments growth of the market.
Bayer AG (Bayer Schering Pharma AG), Danaher Corporation, Becton, Dickinson and Company, Hologic, Inc., Ecolab, Inc., Pfizer, Inc., Steris PLC, Johnson & Johnson, Abbott Laboratories, and Merck Group.
The Urinary Tract Infections (UTI) segment is leading the Global Hospital-Acquired Infection Diagnostics Market by Infection Type in 2021, and would continue to be a dominant market till 2028.
The Molecular Diagnostics segment acquired maximum revenue share in the Global Hospital-Acquired Infection Diagnostics Market by Test Type in 2021; thereby, achieving a market value of $2.2 billion by 2028.
The North America market dominated the Global Hospital-Acquired Infection Diagnostics Market by Region in 2021, and would continue to be a dominant market till 2028.
Our team of dedicated experts can provide you with attractive expansion opportunities for your business.

